Susceptibility of Imidacloprid-Resistant Colorado Potato Beetles to Non-neonicotinoid Insecticides in the Laboratory and Field Trials
Andrei Alyokhin1, Galen Dively2, Megan Patterson3, Matthew Mahoney3, David Rogers3, and John Wollam3
1 Department of Biological Sciences, University of Maine, 5722 Deering Hall, Orono, ME 04469
2 Department of Entomology, University of Maryland, 4112 Plant Sciences Bldg., College Park, MD 20742
3 Bayer CropScience, 2 TW Alexander Dr., Research Triangle Park, NC 27709
This peer-refereed invited review article has been first published by the American Journal of Potato Research (Volume 83, Pages 485-494). The original publication is available at www.springerlink.com. Reproduced with permission of the publisher.
ABSTRACT
Repeated use of neonicotinoid insecticides has resulted in the first reported cases of Colorado potato beetle (Leptinotarsa decemlineata (Say)) resistance to imidacloprid. In the laboratory we determined susceptibility of the imidacloprid-resistant Colorado potato beetles from a population in Southern Maine to other insecticides currently registered for use on potato. This population was about 30-fold resistant to imidacloprid and could not be effectively controlled by its applications. Control mortality was significantly higher for the imidacloprid-resistant larvae than for the susceptible larvae, suggesting that fitness disadvantages may be associated with the resistance trait. Resistant larvae exhibited significantly less mortality than susceptible larvae when exposed to cyfluthrin, carbaryl, azinphosmethyl, and methamidophos. Their susceptibility to oxamyl was also somewhat reduced, although it did provide nearly 100% mortality at the highest concentration tested. Disulfoton was highly toxic to the resistant larvae. Oxamyl killed about 40% of the adults in greenhouse assays with potted potato plants, altered their feeding behavior (fewer adults up on plants), and reduced defoliation by more than 90%. Disulfoton was not lethal to adults, but significantly suppressed their feeding. In field trials with the resistant population, oxamyl and imidacloprid + spinosad provided the best beetle control. Novaluron had no detectable effect on beetle densities. There was little difference between the plots treated with imidacloprid or thiamethoxam and the untreated control. Our results suggest that insecticide rotation may be a valuable option for managing imidacloprid-resistant Colorado potato beetle populations. We also had a good consistency between the results of the Petri dish, greenhouse, and field experiments, indicating that screening under laboratory confinement may be useful when developing initial recommendations to potato growers in areas affected by resistance to neonicotinoids.
Introduction
Colorado potato beetle, Leptinotarsa decemlineata (Say), is the most important insect defoliator of potatoes throughout North America, Europe, and parts of Asia (Weber and Ferro 1994). None of the control techniques developed against this pest during the past 135 years has provided long-term protection for potato crops (Casagrande 1987), and the beetle continues to be a major threat to potato production. High fecundity, a diverse and flexible life history, and a remarkable ability to develop insecticide resistance make Colorado potato beetle management a challenging task.
Colorado potato beetle propensity for developing insecticide resistance is very costly for potato growers (Grafius 1997). Many of the Colorado potato beetle's host plants (family Solanaceae) contain high concentrations of toxins, which may contribute to the beetle's ability to adapt to insecticides (Ferro 1993). The first instance of beetle resistance to synthetic organic pesticides was noted for DDT in 1952 (Quinton 1955). Since then, failures have been reported for virtually all classes of insecticides (Hofmaster et al. 1967; Forgash 1985; Boiteau 1988; Ioannidis et al. 1991; Stewart et al. 1997; Noronha et al. 2001; Stankovic et al. 2004). In some cases, a new insecticide lost its efficiency after one year (e.g., endrin) or even during the first year of use (e.g., oxamyl) (Forgash 1985).
Resistance problems reached critical levels in the early 1990s, when many potato growers completely ran out of chemical control options. The situation improved dramatically after neonicotinoid insecticides became commercially available in 1995. Novel mode of action made this class of insecticides an effective tool in areas where the beetles had developed resistance to other chemicals. Long residual activity and low mammalian toxicity provided additional incentive for their wide adoption by commercial growers.
Imidacloprid was the first neonicotinoid to appear on the market and the most widely used neonicotinoid insecticide on potatoes. Growers usually apply Admire (Bayer CropScience, Research Triangle Park, NC) to the whole field at planting. This maximizes plant coverage and significantly increases insecticide persistence in potato foliage. Unfortunately, whole-field systemic applications also create strong selection pressure on insect populations towards developing resistance to this compound. Most Colorado potato beetle populations are still susceptible to neonicotinoid insecticides, including imidacloprid. Moreover, neonicotinoids still remain the most effective and reliable chemical option available to the potato grower. However, the first instances of resistance to imidacloprid have been already reported from commercial potato farms in New York, Delaware, and southern Maine (Zhao et al. 2000; Olson et al. 2000; personal observations). Unless appropriate actions are taken to minimize exposure and subsequent selection pressure to vulnerable populations, it is reasonable to expect eventual failure of neonicotinoid compounds for control of the Colorado potato beetle, similar to the failure of other classes of insecticides.
Insecticide rotation to different modes of action is one of the most commonly recommended resistance management techniques (IRAC 2005; NPC 2005). However, Colorado potato beetles are commonly resistant to multiple chemicals belonging to different insecticide classes (Ioannidis et al. 1991). Furthermore, resistance mechanisms may be highly diverse even within a relatively narrow geographical area (Ioannidis et al. 1991). Therefore, selection of appropriate rotation chemicals may be a rather challenging task due in part to scarcity of suitable, economically viable alternatives and lack of sufficient understanding of resistance mechanisms by growers. Determining susceptibility of the imidacloprid-resistant Colorado potato beetles to other insecticides currently registered for use on potatoes was the primary objective of the present study.
Materials and Methods
Beetle origins and study site. The Colorado potato population used in this study originated from several commercial potato fields in Fryeburg, Oxford County, Maine (44º01’53’’ N, 70º58’01’’ W). The fields were located within a relatively small (8 km long by 2.4 km wide) valley that is isolated from other potato growing areas (at least 40 km to the nearest commercial potato farm). Most potato fields in the area were rotated annually or biennially with corn and green manure crops. Climate in the valley is warmer compared to other potato-growing areas in Maine and allows for two Colorado potato beetle generations in most years.
According to the growers, potato beetles in the valley have had a long history of insecticide resistance. Pyrethroids, carbamates, and organophosphates were used for beetle control before imidacloprid had become available, but eventually lost their efficiency even when used as tank mixtures of several insecticides with different chemistries. In-furrow applications of imidacloprid provided excellent control for the first four years of use, but the residual efficacy started to decline in subsequent years. Hotspots of highly neonicotinoid resistant beetles first appeared in 2002, and valley-wide decline in beetle control was observed in 2003.
Baseline susceptibility to imidacloprid. Baseline susceptibility was determined for the Maine population described above using a diet incorporation assay (Olson et al. 2000). Post-diapause adults were collected from a single location in 2003 and two different locations in 2004 (a total of three samples from three different locations within 5 km from each other). Approximately 200 beetles per sample were captured, placed in Styrofoam boxes that contained frozen ice packs, and shipped overnight to the University of Maryland Wye Research and Education Center, Queenstown, MD. Upon receipt, they were kept on caged potato plants and checked daily for egg laying. Egg masses were then placed in Petri dishes and kept in an environmental chamber at 24ºC and a photoperiod of 16:8 (L:D) h until hatching. Neonate larvae were allowed to feed on their egg chorions until they began to disperse and then were transferred to fresh potato leaves.
After 5 h of feeding on leaves, first instars were placed in plastic bioassay trays containing a potato-based agar Colorado potato beetle diet (Olson et al. 2000). Each tray consisted of six columns of five separate wells 1.5 cm by 2 cm by 1.5 cm deep. Ten larvae were placed in a well. Each column contained diet incorporated with one of five imidacloprid concentrations and an untreated control. The concentration series was selected to bracket the 50% lethal level, allowing a full concentration-mortality response. At least three replicates of bioassay trays (30 larvae/concentration) were conducted for each sample. The trays were maintained in an environmental chamber at 24ºC and a photoperiod of 16:8 (L:D) h. After 48 h, individual larvae were nudged with a probe or placed on their dorsal sides and recorded as alive if they flipped over or advanced one step in a coordinated way. Tests were repeated if control mortality exceeded 10%. Original data were discarded if there were no signs of cannibalism, and/or the lowest concentration resulted in higher mortality than the control. Standard probit analysis (PROC PROBIT, SAS Institute 1999) was used to model the concentration-mortality responses to estimate concentrations killing 50% of the exposed population (LC50).
In addition, a beetle population from a potato field in Kimberly, Twin Falls County, Idaho (42º31’43’’ N, 114º21’57’’ W) was reared as a susceptible reference (KI) and assayed following the same protocol as described above. That population was exposed to pyrethroids three times, to carbofuran twice, to a mixture of cyfluthrin and imidacloprid (Leverage®, Bayer CropScience, Research Triangle Park, NC), and to foliar formulation of imidacloprid (Provado®, Bayer CropScience, Research Triangle Park, NC) twice during the past 5 years. All insecticides provided an excellent Colorado potato beetle control as reported by the grower.
Larval susceptibility to non-neonicotinoid insecticides in the laboratory. This part of the study was conducted to predict field efficacy of cyfluthrin (95.8% technical grade, Bayer CropScience, Research Triangle Park, NC), disulfoton (98.5% technical grade, Bayer CropScience, Research Triangle Park, NC), azinphosmethyl (94% technical grade, Bayer CropScience, Research Triangle Park, NC), methamidophos (75% technical grade, Bayer CropScience, Research Triangle Park, NC), carbaryl (99% technical grade, Bayer CropScience, Research Triangle Park, NC), and oxamyl (99% technical grade, DuPont, Wilmington, DE), against the imidacloprid-resistant Colorado potato beetles. Cyfluthrin is a pyrethroid, disulfoton, azinphosmethyl, and methamidophos are organophosphates, and carbaryl and oxamyl are carbamates. Eggs laid by the field-collected imidacloprid-resistant beetles (see above) were used to establish a laboratory colony. To maintain the level of resistance, the colony was selected for one generation by feeding second instars for two days on diet treated with a 90% lethal concentration of imidacloprid, and then used in the assays. In addition, the susceptible KI beetle population (F1 generation relatively to field-collected beetles) was reared to provide a reference for comparison.
Bioassays were conducted concurrently on both populations by exposing first instars to insecticide residue on filter paper in Petri dishes (Heim et al. 1990; Olson et al. 2000). Filter paper disks (5.5 cm diameter; Fisher Scientific, Raleigh, NC) were treated with 0.5 solution of tested insecticides in acetone. Previous studies using the same exposure test showed that percent mortality caused by discriminating concentrations of 0.1 µg/µl of esfenvalerate, 2.5 µg/µl of azinphosmethyl, and 10 µg/µl of oxamyl were directly correlated to percent control in the field with high-labeled rates (Ellis 1989; Olson et al. 2000; Dively unpublished). As a general rule, economically acceptable control using esfenvalerate, azinphosmethyl, or oxamyl is likely if a CPB population exhibits >80% mortality in exposure bioassays using these discriminating concentrations. Since no correlative data are available to relate bioassay results with field performance of chemicals tested in the present study, discriminating concentrations were estimated by multiplying the known discriminating concentrations of either esfenvalerate, azinphosmethyl, or oxamyl times the ratio of active ingredient amounts in the high-labeled rates of matching chemicals within the same mode of action. Esfenvalerate was used to determine discriminating concentration for cyfluthrin, azinphosmethyl was used for disulfoton and methamidophos, and oxamyl was used for carbaryl. Using this approach, discriminating concentrations of 30 µg/µl for disulfoton, 0.04 µg/µl for cyfluthrin, 20 µg/µl for carbaryl, and 10 µg/µl for methamidophos were established. In addition, one or two lower concentrations diluted in 5-fold steps were used for each chemical to bracket a range of mortalities.
Egg masses were collected from potted potato plants in rearing cages of each population (imidacloprid-resistant and -susceptible) and reared until hatching. Larvae were collected for bioassay as they began to disperse. Ten to twenty larvae were randomly selected from a pooled sample obtained from each population and placed in each Petri dish. Mortality was assessed after 24 hours as described above.
For the resistant population, between nine and sixteen replicate tests of each discriminating concentration and an acetone control were conducted for each chemical. The susceptible KI population had fewer reproductive adults in colony and thus eggs were limited. Therefore, only four replicates of each concentration and acetone control were conducted. Also, if mortality in the susceptible strain exposed to a given concentration of a given chemical exceeded 85% (see above), it was not exposed to the higher concentrations of the same chemical.
Mortality of imidacloprid-resistant beetles exposed to discriminating concentrations was compared among different insecticides. To normalize its distribution, the data were transformed using arcsine (Öx) transformations (Zar 1999). Effects of decreasing concentrations on mortalities of imidacloprid-resistant beetles were then further analyzed for each chemical by logistic regression (PROC LOGISTIC, When both strains were tested, the difference between imidacloprid-resistant beetles and susceptible beetles exposed to the same concentration of the same chemical were analyzed by median two-sample tests (PROC NPAR1WAY, SAS Institute 1999). We chose to use non-parametric tests because transforming the data failed to normalize its distribution, and relatively small sample size would have made parametric tests insufficiently powerful (Zar 1999).
Adult susceptibility to soil-applied systemic insecticides in the greenhouse. The objective of this part of the study was to determine if oxamyl (VydateÒ, DuPont, Wilmington, DE), and disulfoton (DisystonÒ, Bayer CropScience, Research Triangle Park, NC) insecticides could be used in place of neonicotinoids to provide early season protection against overwintered adult beetles. One potato seed piece with active eyes was placed approximately 7.5 cm deep in 15 cm plastic pots filled with a standard potting soil. Pots were maintained in the greenhouse until potato shoots appeared at the surface. At this time, 25 ml of 1 mg a.i./ml oxamyl solution or 25 ml of 3 mg a.i./ml disulfoton solution per pot was applied as a water solution over the soil surface. These rates were equivalent to the highest label rate of each chemical, assuming that one potted plant represented 30 cm linear row. Treated pots were then placed into the center of 25 cm pots with extra soil placed beneath and around the smaller pot. An additional 5 cm layer of soil was placed over the treated pot to simulate a hilling operation to incorporate the chemical. The larger pots were then held in the greenhouse until potato plants emerged again, at which time four adult beetles were enclosed in a cardboard container that was inserted in the soil over the young shoots. Six replicates of potted plants treated with each chemical and an untreated control were evaluated. Adult mortality and adult activity were recorded after 3 and 5 d. Adult activity on each date was determined using a single “snap-shot” observation, during which the percent of surviving beetles that were moving, feeding, and mating was recorded. Percent defoliation was estimated after 5 and 20d. Differences among the treatments were compared by repeated measures ANOVA (PROC GLM, SAS Institute 1999). To normalize its distribution, the data were transformed using arsine (Öx) transformations (Zar 1999).
Field efficacy trials. We tested effects of seven different insecticidal treatments on imidacloprid-resistant Colorado potato beetles. The treatments were as follows: imidacloprid (Admire®), thiamethoxam (Platinum®, Syngenta Crop Protection, Greensboro, NC), imidacloprid (Admire®) followed by spinosad (SpinTor®, Dow AgroSciences, Indianapolis, IN), disulfoton (DisystonÒ), novaluron (RimonÒ, Chemtura, Middlebury, CT), oxamyl (VydateÒ), and untreated control. The experiment was arranged in a complete randomized block design, with seven treatment plots randomized within three blocks. Each plot was 9 m long and 3.6 m (four rows) wide. The plots within a block were separated from each other by 1.8 m of fallow ground, and the blocks were separated by 6 m of fallow ground. The experimental plots were separated from the adjacent commercial potato field by 3.6 m wide strip of fallow ground. The commercial field was treated with disulfoton in furrow at planting, and then with alternating sprays of oxamyl and spinosad throughout the growing season. It was planted to potatoes in the preceding year and had a history of being treated with imidacloprid and spinosad during the five years before our study.
Potatoes at the experimental plots were planted on May 21, 2004. Imidacloprid (280 g ai/ha), thiamethoxam (140 g ai/ha), and disulfoton (840 g ai/ha) were applied in furrow at planting. Oxamyl (1060 g ai/ha) and novaluron (87 g ai/ha) were applied on June 17, June 30, and July 31. Spinosad (75 g ai/ha) was applied on June 30, and July 31. All applications were made using CO2-pressured hand sprayer (two nozzles, one nozzle over one row) and intended to simulate commercial applications. In-furrow sprays were made using flat fan nozzles at 83 kPa and flow rate of 79 l/ha. Foliar sprays were made using hollow cone nozzles at 241 kPa and flow rate of 337 l/ha.
Plots were scouted weekly throughout the growing season. Twenty-five potato plants were randomly selected from all four rows within each plot for weekly visual examinations. The number of Colorado potato beetles on each selected plant was recorded. Different Colorado potato beetle life stages (eggs, small larvae, large larvae, and adults) were recorded separately. First and second instars were recorded as small larvae, while third and fourth instars were recorded as large larvae. The data were analyzed by repeated measures ANOVA (PROC GLM, SAS Institute 1999), followed by Tukey mean separation tests.
Percent defoliation was visually estimated on August 4 and August 27. To minimize human error, estimations were “blind” (without reference to treatment) and two readings were taken at each plot on each date. The readings were later pooled for the statistical analyses. The data were analyzed by repeated measures ANOVA (PROC GLM, SAS Institute 1999), followed by Tukey mean separation tests. To normalize its distribution, the data were transformed using arcsine (Öx) transformations (Zar 1999).
Tubers from one outside and one middle row of each plot were harvested using a tractor-mounted potato digger on October 1 and weighed in the field. The data were analyzed by ANOVA (PROC GLM, SAS Institute 1999). Possible connection between yields, beetle densities, and defoliation levels was tested using Pearson’s correlation analysis (PROC CORR, SAS Institute 1999).
Results
Baseline susceptibility to imidacloprid. Beetles from all three samples collected in southern Maine were highly resistant to imidacloprid (Table 1). For reference, initial Colorado potato beetle baseline sensitivity to imidacloprid for US populations, excluding Long Island, NY was established as LC50 of 0.39 ppm (Olson et al. 2000). Although there was a considerable variation within each sample, mean LC50 values were 20 to 40 times higher for the southern Maine imidacloprid-resistant population compared to the KI susceptible reference (significantly different, P<0.05, based on non-overlap of confidence intervals)(Heim et al. 1992).
Table 1. Baseline susceptibility of the tested Colorado potato beetle populations to imidacloprid.
Year |
Origin |
No. of Larvae Tested |
LC50 (ppm) |
Slope +/- SEM |
Goodness-of-Fit |
|
|
|
|
|
|
χ2 |
P |
2003 |
Idaho |
1408 |
0.33 (0.2-0.5) |
1.74 ± 0.27 |
75.11 |
<0.0001 |
2003 |
Southern Maine |
1515 |
12.19 (8.9-19.5) |
1.08 ± 0.15 |
11.42 |
0.0096 |
2004 |
Southern Maine |
734 |
8.17 (1.55-18.99) |
1.76 +/-0.35 |
7.45 |
0.8776 |
2004 |
Southern Maine |
740 |
6.71 (1.13-15.18) |
1.78 +/-0.32 |
14.10 |
0.3665 |
Larval susceptibility to non-neonicotinoid insecticides in the laboratory. Mortality of imidacloprid-resistant Colorado potato beetles exposed to discriminating doses of different insecticides was significantly different among the treatments (df=6, 119, F=77.22, P<0.0001)(Fig. 1). Not surprisingly, all tested insecticides increased beetle mortality. Of those, disulfoton, azinphosmethyl, and oxamyl killed the highest proportion of exposed larvae. Methamidophos was less toxic, but still resulted in the death of ca. 80% of the tested larvae. Exposure to carbaryl caused relatively low mortality, and effect of cyfluthrin was intermediate between carbaryl and methamidophos.
Decrease in the exposure dose led to a significant decrease in beetle mortality for cyfluthrin (χ2= 13.94, P=0.0002), azinphosmethyl (χ2= 70.50, P<0.0001), methamidophos (χ2= 39.97, P<0.0001), and oxamyl (χ2= 51.42, P<0.0001). No such effect was observed for disulfoton (100% mortality regardless of its dose) and carbaryl (χ2= 1.38, P=0.2399)(Fig. 1).
Control mortality was significantly higher for the imidacloprid-resistant larvae than for the susceptible larvae (P=0.0289). The opposite generally became true when the beetles were exposed to insecticides (Fig. 1). The differences between the strains were statistically significant (P<0.05) in all the cases except for methamidophos at 2 mg/ml, carbaryl, and oxamyl.
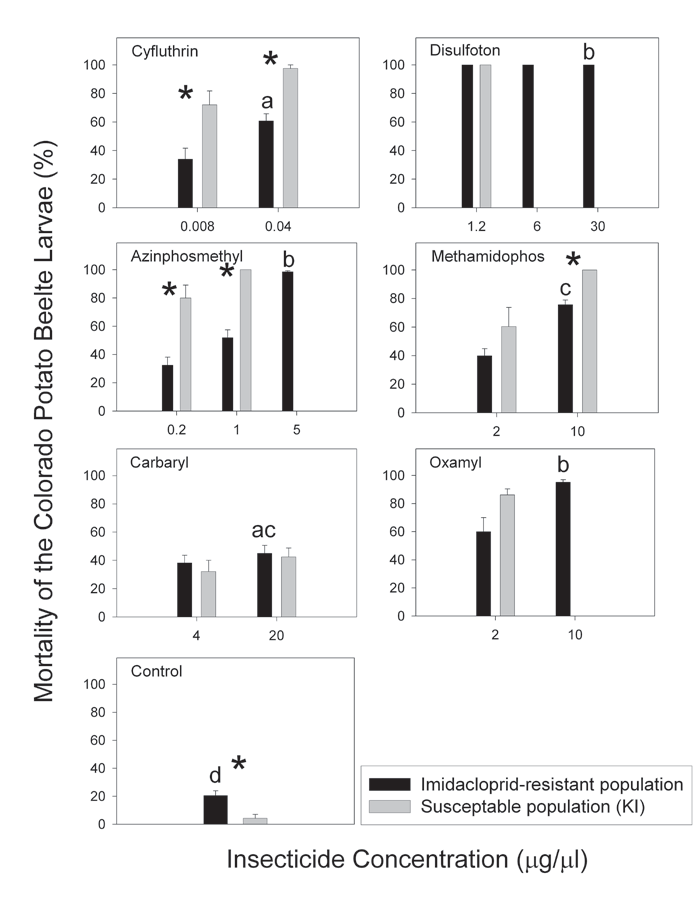
Figure 1. Mortality of imidacloprid-resistant and susceptible Colorado potato beetle larvae exposed to non-neonicotinoid insecticides in Petri dish bioassays. Bars followed by the same letters indicate mortalities caused by discriminating doses of different insecticides that were not significantly different from each other (P>0.05; see text for details). Asterisks indicate statistically significant differences (P<0.05) between imidacloprid-resistant and susceptible strains.
Adult susceptibility to soil-applied systemic insecticides in the greenhouse. Treatment had a highly significant effect on beetle mortality (df=2, 15, F=14.95, P=0.0003), with most dead beetles observed on potato plants treated with oxamyl (Fig. 2A). Percent surviving beetles that were active on plants followed a similar trend (Fig. 2B), and the difference between the treatments was also significant (df=2, 15, F=3.79, P=0.0466). Defoliation was strongly affected by the treatment (df=2, 15, F=35.53, P=0.0003). Interestingly, although disulfoton-treated plants had similar beetle density and activity with control plants, they were less defoliated (Fig. 2C).
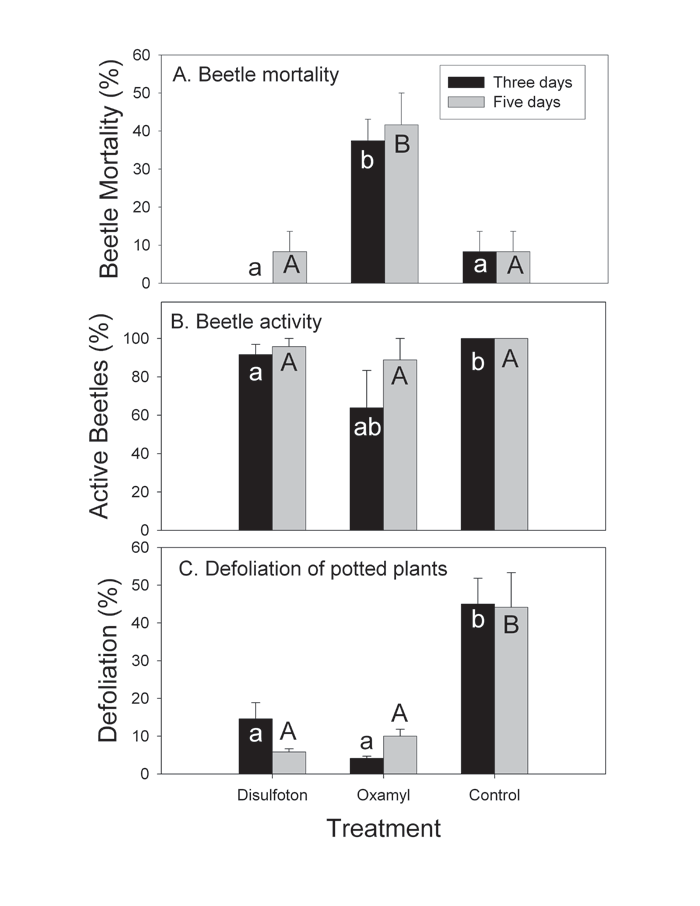
Figure 2. Effects of soil applications of disulfoton and oxamyl to potted plants in the greenhouse on Colorado potato beetle adults: (A) beetle mortality; (B) beetle activity (percent surviving beetles that were moving, feeding, and mating); (C) defoliation of potted plants. Bars followed by the same letters were not significantly different from each other (P>0.05). Error bars indicate standard errors.
Field efficacy trials. Treatment had a highly significant effect on the densities of all life stages except egg masses (Fig. 3). Populations on plots treated by imidacloprid and thiamethoxam were not significantly different from the populations on the untreated control plots, while imidacloprid + spinosad and oxamyl significantly reduced beetle densities. Disulfoton and novaluron failed to reduce beetle populations.
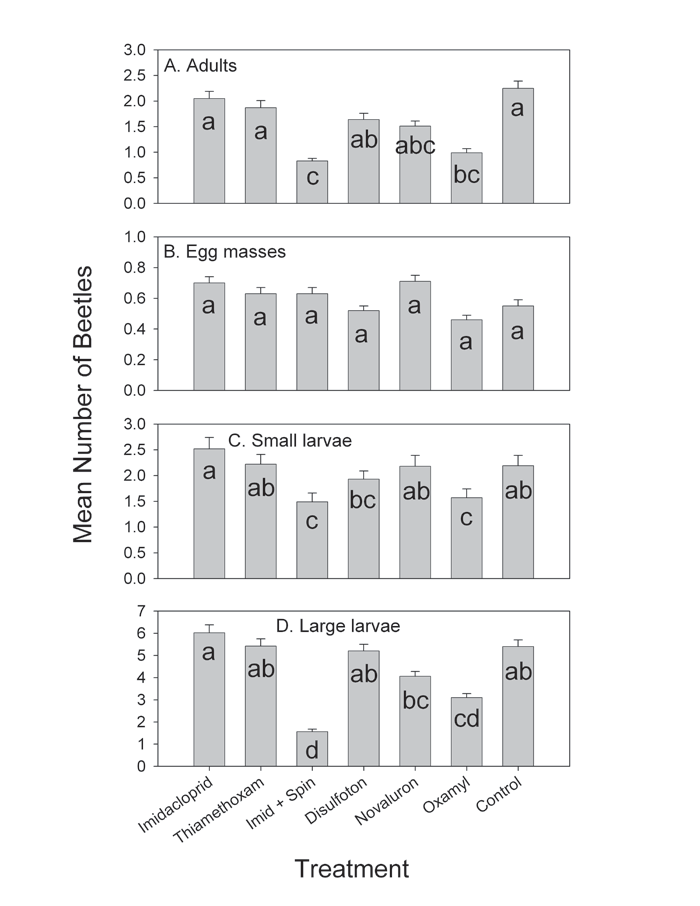
Figure 3. Overall seasonal densities of the Colorado potato beetle life stages on experimental field plots. Error bars indicate standard errors. Bars followed by the same letters were not significantly different from each other (P>0.05). Error bars indicate standard errors.
Defoliation was correlated with Colorado potato beetle densities on the respective treatment plots (Fig. 4; Table 2). Treatment effect was statistically highly significant (F=3.28; df=6,35; P=0.0115), and so was the interaction between treatment and time (F=3.59; df=6,35; P=0.0071). On 4 August, plots sprayed with oxamyl and spinosad sustained the least amount of damage. On 24 August, the amount of defoliation increased on all plots and the difference became statistically insignificant (Fig. 4). Disulfoton, novaluron, and neonicotinoids had no impact on potato defoliation.
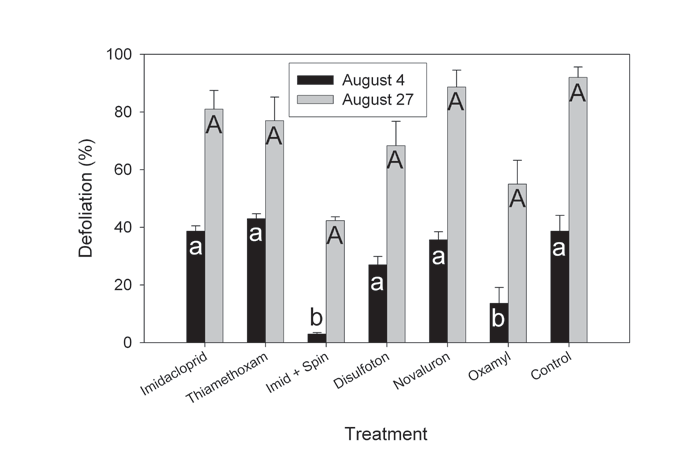
Figure 4. Defoliation of potato plants on insecticide trial plots conducted on a commercial potato farm in southern Maine that has an imidacloprid-resistant Colorado potato beetle population. Error bars indicate standard errors. Bars followed by the same letters were not significantly different from each other (P>0.05). Error bars indicate standard errors.
Yields ranged from 30,732 ± 2,772 kg/ha (mean ± SE) on the control plots to 44,620 ± 3,631 kg/ha on the plots treated with imidacloprid and spinosad. Yields were correlated with densities of all beetle life stages except egg masses, as well as with defoliation indices (Table 2). However, the difference among the treatments was not statistically significant (F=2.49; df=6,14; P=0.0745).
Discussion
Both LC50 values obtained in laboratory assays, as well as Colorado potato beetle densities on experimental plots confirmed grower observations of imidacloprid failure on the affected farms. Imidacloprid still had some deleterious effects on the beetles. However, the levels and length of control expressed were clearly unacceptable for commercial growers, with 80% defoliation by the end of the season. Field performance of imidacloprid and thiamethoxam was very similar, suggesting a considerable degree of cross-resistance between the two neonicotinoids.
The imidacloprid-resistant population was susceptible to a number of non-neonicotinoid insecticides. Petri dish bioassays revealed fairly high mortality of beetle larvae exposed to disulfoton, azinphosmethyl, and oxamyl. This suggests that insecticide rotation may be a valuable option for managing imidacloprid-resistant populations. However, mortality of resistant larvae was invariably lower than mortality of susceptible larvae exposed to the same insecticide concentrations (Fig. 1). Imidacloprid-resistant population used in the present study had a long history of exposure (and subsequent resistance) to a variety of chemical classes. Therefore, it is likely that this population had a fairly high frequency of alleles conferring resistance to other classes of insecticides prior to its exposure to imidacloprid. Our finding suggests that there might be reinvigoration of resistance development to at least some of the chemicals used as an alternative to imidacloprid, despite their long-term removal from population exposure. Also, it is worth noticing that there was a considerable variation in toxicity of different insecticides belonging to the same chemical class. For example, exposure to oxamyl resulted in high larval mortality, while carbaryl was essentially a failure. Similarly, disulfoton performed better than azinphosmethyl, and especially than methamidophos. Knowing the history of insecticide exposure and susceptibility to individual insecticides for individual Colorado potato beetle populations is essential for achieving good levels of beetle control. Simply replacing one class of chemicals with another is not necessarily going to provide long-term protection of a potato crop from beetle damage.
In the absence of exposure to insecticides, mortality was more than three times higher for the imidacloprid-resistant larvae compared to the susceptible larvae (Fig. 1G). Since cohorts of both populations were reared under the same conditions, this suggests that fitness disadvantages may be associated with the imidacloprid (neonicotinoid)-resistance trait. Similar decreased relative fitness of resistant genotypes in insecticide-free environments was observed in the Colorado potato beetle strains resistant to several insecticides (Argentine et al. 1989; Trisyono and Whalon 1997; Alyokhin and Ferro 1999). This puts resistant genotypes at a selective disadvantage in the absence of insecticides, favoring population reversion to susceptibility (Tabashnik 1994; Caprio 1998; Carrière and Tabashnik 2001). Therefore, resistance management techniques that allow for the existence of a sufficiently large susceptible population, such as refugia and insecticide rotations, may delay evolution of imidacloprid resistance in the Colorado potato beetle.
When defoliation of emerging potato plants by overwintered adults early in the season is a concern, disulfoton and oxamyl may help to address the problem. In greenhouse studies, oxamyl had a strong negative effect on the exposed beetles (Fig. 2). However, it is not labeled for in-furrow applications in Northeast and mid-Atlantic United States. If feeding adults eliminate potato sprouts soon after they appear above ground (which is possible when beetle densities are very high), using oxamyl would require very precise timing. Disulfoton appeared to have mostly sublethal effect on the adult beetles. Those effects were strong enough to result in lower defoliation of potted plants compared to the untreated control plants (Fig. 2C), but not particularly pronounced in the field (Fig. 3). Disulfoton can be applied in furrow at planting, making it potentially useful for protecting emerging young plants from being eliminated by overwintered adults. However, it should be used only when there is a good reason to expect really high populations of overwintered adults. A history of exposure to organophosphates in most beetle populations, together with already low toxicity of disulfoton observed in our study suggests a considerable additional threat of resistance development in beetle populations.
Spinosad provided good control of imidacloprid-resistant beetles that were feeding on imidacloprid-treated potato plants. This suggests that foliar spinosad applications may be used to supplement control when growers first notice failure of in-furrow applied imidacloprid. They are also likely to be a valuable component of insecticide rotation programs designed to delay the evolution of insecticide resistance. In the same time, it is important to remember that although Insecticide Resistance Action Committee places spinosad in a separate MOA class than neonicotinoids (IRAC 2005), both have a fairly similar mode of action (nicotinic acetylcholine receptor agonists). Therefore, spinosad should not be relied upon as a single alternative to neonicotinoids, and should be rotated with chemicals that have a different mode of action.
Novaluron was somewhat of a disappointment, providing a relatively poor larval control in our field trial. However, being an inhibitor of chitin biosynthesis this compound has a distinct advantage of having a novel mode of action. As such, it might still be worth being included in resistance management plans. In the areas that have more than one beetle generation per year, numbers of the first summer generation beetles may be dramatically reduced by applications of more efficient insecticides (e.g., spinosad and oxamyl). The less abundant second generation may potentially be kept below damaging levels by timely applications of novaluron. However, more testing is required before developing specific recommendations, and those are likely to vary in different growing areas.
Our study was conducted with a single imidacloprid-resistant population from a relatively isolated geographic location. Mechanisms and levels of resistance in the Colorado potato beetle populations may be highly diverse even within a relatively narrow geographical area (Ioannidis et al. 1991). Therefore, results of this specific study will be most applicable to fields with similar histories of insecticide use and resistance. However, we believe that they provide a good early insight into the problem. Also, there was good consistency between the results of the Petri dish, greenhouse, and field experiments. This indicates that screening under laboratory confinement may be useful when developing initial recommendations to potato growers in areas impacted by neonicotinoids resistance.
Acknowledgments
We thank Gary Sewell, Earle Towle, Amy Miller, and Terry Patton for technical assistance. This is Publication No. 2893 of the Maine Agricultural and Forest Experiment Station.
References
Alyokhin AV, and DN Ferro. 1999. Relative fitness of Colorado potato beetle (Coleoptera: Chrysomelidae) resistant and susceptible to the Bacillus thuringiensis Cry3A toxin. J Econ Entomol 92:510-515.
Argentine JA, JM Clark, and DN Ferro. 1989. Relative fitness of insecticide-resistant Colorado potato beetle strains (Coleoptera: Chrysomelidae). Environ Entomol 18:705-710.
Boiteau G. 1988. Control of the Colorado potato beetle, Leptinotarsa decemlineata (Say): learning from the Soviet experience. Bull Entomol Soc Can 20:9-14.
Caprio MA. 1998. Evaluating resistance management strategies for multiple toxins in the presence of external refuges. J Econ Entomol 91:1021-1031.
Carrière Y, and BE Tabashnik. 2001. Reversing insect adaptation to transgenic insecticidal plants. Proc R Soc Lond B. 268:1475-1480.
Casagrande RA. 1987. The Colorado potato beetle: 125 years of mismanagement. Bull Entomol Soc Am 33:142-150.
Ellis FW. 1989. Baseline levels and factors associated with insecticide resistance in Colorado potato beetle populations in Maryland. M.S. thesis, University of Maryland, College Park, MD.
Ferro DN. 1993. Potential for resistance to Bacillus thuringiensis: Colorado potato beetle (Coleoptera: Chrysomelidae) - a model system. Am. Entomol. 39:38-44.
Forgash AJ. 1985. Insecticide resistance in the Colorado potato beetle. In: DN Ferro and RH Voss (eds), Proceedings of the Symposium on the Colorado Potato Beetle, 17th International Congress of Entomology. Massachusetts Experiment Station, University of Massachusetts, Amherst, MA, pp. 33-53.
Grafius E. 1997. Economic impact of insecticide resistance in the Colorado potato beetle (Coleoptera: Chrysomelidae) on the Michigan potato industry. J Econ Entomol 90:1144-1151.
Heim DC, GG Kennedy, and JW Van Duyn. 1990. Survey of insecticide resistance among North Carolina Colorado potato beetle (Coleoptera: Chrysomelidae) populations. J Econ Entomol 83:1229-1235.
Heim DC, GG Kennedy, FL Gould, and JW Van Duyn. 1992. Inheritance of fenvalerate and carbofuran resistance in Colorado beetles – Leptinotarsa decemlineata (Say) – from North Carolina. Pestic Sci 34:303-311.
Hofmaster RN, RL Waterfield, and JC Boyd. 1967. Insecticides applied to the soil for control of eight species of insects on Irish potatos in Virginia. J Econ Entomol 60:1311-1318.
Insecticide Resistance Action Committee (IRAC). 2005. IRAC mode of action classification, version 5.1. http://www.iraconline.org/documents/moa/MoAv5_1.pdf
Ioannidis PM, E Grafius, and ME Whalon. 1991. Patterns of insecticide resistance to azinphosmethyl, carbofuran, and permethrin in the Colorado potato beetle (Coleoptera: Chrysomelidae). J Econ Entomol 84:1417-1423.
Quinton RJ. 1955. DDT-resistant Colorado potato beetles? Proc North Centr Entomol Soc Am 9:94-95.
National Potato Council (NPC). 2005. Neonicotinoid Insecticides: A Grower Approach to Resistance Management of Colorado Potato Beetle and Green Peach Aphid in Potato. http://www.nationalpotatocouncil.org
Noronha C, GM Duke, JM Chinn, and MS Goettel. 2001. Differential susceptibility to insecticides by Leptinotarsa decemlineata (Coleoptera: Chrysomelidae) populations from western Canada. Phytoprotection 82:113-121.
Olson ER, GP Dively, and JO Nelson. 2000. Baseline susceptibility to imidacloprid and cross resistance patterns in Colorado potato beetle (Coleoptera: Chrysomelidae) populations. J Econ Entomol 93:447-458.
SAS Institute, Inc. 1999. SAS OnLine Doc, Version 8. Cary, North Carolina.
Stankovic S, A Zabel, M Kostic, B Manojlovic, and S Rajkovic. 2004. Colorado potato beetle (Leptinotarsa decemlineata (Say)) resistance to organophosphates and carbamates in Serbia. J Pest Sci 77:11-15.
Stewart JG, GG Kennedy, and AV Sturz. 1997. Incidence of insecticide resistance in populations of Colorado potato beetle, Leptinotarsa decemlineata (Say) (Coleoptera: Chrysomelidae), on Prince Edward Island. Can Entomol 129:21- 26.
Tabashnik BE. 1994. Evolution of resistance to Bacillus thuringiensis. Annu Rev Entomol 39:47-79.
Trisyono A, and ME Whalon. 1997. Fitness costs of resistance to Bacillus thuringiensis in Colorado potato beetle (Coleoptera: Chrysomelidae). J Econ Entomol 90:267-71.
Weber DC, and DN Ferro. 1994. Colorado potato beetle: diverse life history poses challenge to management. In: GW Zehnder, RK Jansson, ML Powelson, and KV Raman (eds), Advances in Potato Pest Biology and Management. APS Press, St. Paul, pp. 54-70.
Zar JH. 1999. Biostatistical Analysis, 4th Edition. Prentice Hall, Upper Saddle River, New Jersey.
Zhao J-Z, BA Bishop, and EJ Grafius. 2000. Inheritance and synergism of resistance to imidacloprid in the Colorado potato beetle (Coleoptera: Chrysomelidae). J Econ Entomol 93:1508–1514.